Mesenchymal Stem Cells: A Novel Therapy for Type 2 Diabetes Mellitus

Type 2 Diabetes Mellitus (T2DM) is a common chronic metabolic disorder caused by impaired regulation of blood sugar level and reduction of insulin-sensitive tissues response to elevation of insulin in blood. The organs involved in T2DM pathogenesis include the pancreas (β-cells and α-cells), which is responsible for producing insulin; the skeletal muscle, and adipose tissue and liver, which are insulin sensitive tissues, and; nerves, blood vessels, heart, kidneys, eye, and brain, which are the most vulnerable organs to high glucose levels in blood.
While individual genetic predisposition to T2DM has strong evidence, epidemiological studies suggest that many cases of T2DM can be prevented or delayed by improving the main modifiable risk factors (obesity, low physical activity, and unhealthy diet). In particular, obesity (body-mass index [BMI]≥30 kg/m2) is the strongest risk factor for T2DM due to metabolic abnormalities occurring in obese people resulting in IR (insulin resistance).
Likewise, a sedentary lifestyle is another risk factor for T2DM development. It was shown that only 2-3h of walking per week could reduce development of T2DM by 56%. There are three mechanisms by which physical activity delays the T2DM onset. First, the contraction of skeletal muscle cells increases blood flow into the muscle and enhances glucose uptake from plasma. Moderate-intensity exercise has been estimated to improve glucose uptake by 40%. Second, physical activity reduces the fat depositions, which is a risk factor of IR development. And third, regular exercise and moderate physical activity enhance the production of anti-inflammatory cytokines, antioxidants (such as glutathione, which lead to a long-term reduction in free radical levels), and irisin (an exercise-regulated myokine and adipokine). It has been shown that T2DM patients with cardiovascular disease (CVD) have significantly lower serum irisin than non-CVD patients, and low levels of serum irisin are associated with 1.6 times increased risk of CVD incidence in T2DM patients.
T2DM typically starts with a resistance to insulin of skeletal muscle, adipose and liver tissue, that worsens over time. In an excessive nutritional state, characteristics of obesity, hyperglycemia, and hyperlipidemia are present, favoring insulin resistance and chronic inflammation development. These circumstances trigger compensating mechanisms of insulin overproduction by pancreatic beta cells, followed by beta cell exhausting, loss of signaling cell to cell integrity, and finally, beta cell failure. When most of the beta cells are damaged, the deficiency of endogenous insulin secretion occurs, thereby requiring insulin administration.
>
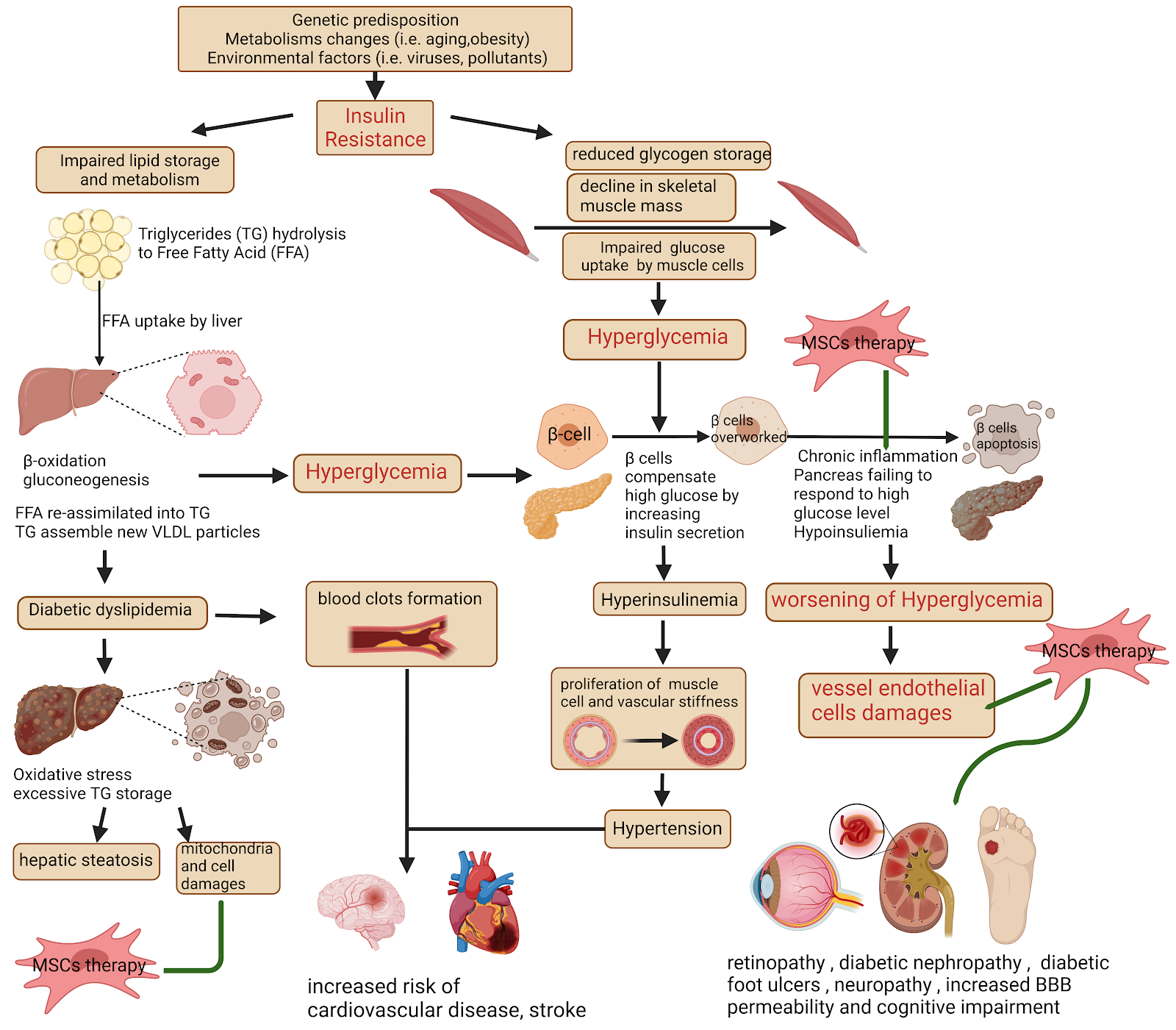
Picture was created with BioRender
Skeletal muscle IR development is the most important non pancreatic origin in the T2DM onset. Insulin normally stimulates muscle glycogen synthesis by enhancing glucose uptake from plasma. Genetic mutations, defective epigenetic regulation of genes occurred during lifetime, and environmental factors (obesity, sedentary lifestyles, unhealthy diets) make the glucose uptake by muscle tissue insufficient, thereby causing hyperglycemia.
Relatively, obesity increases immune cell infiltration and secretion of proinflammatory molecules in adipose tissue resulting in skeletal muscle inflammation, impaired myocyte metabolism, and IR development through paracrine effects. An inadequate response of adipose tissue to insulin stimulation or adipose tissue IR leads to impaired suppression of lipolysis, lowering glucose uptake, and enhanced FFA (free fatty acids) release from the intracellular TG (triglycerides) stores of adipocytes into plasma even in the presence of high insulin levels.
When excessive amounts of FFA in the blood are accumulated in the liver by hepatocytes, FFA can be directed to the mitochondria and undergo β-oxidation, or be re-assimilated into TG leading to hepatic steatosis, or promote gluconeogenesis, resulting in a worsening of hyperglycemia. Excessive TG accumulation in the liver also leads to the assembly of new VLDL particles that could not be properly processed, contributing to bad cholesterol metabolism and atherosclerosis development. In fact, one of the hallmark traits of T2DM is a characteristic dyslipidaemic profile consisting of elevated TG, TG-rich lipoproteins, small dense LDLs, and reduced HDL levels.
Likewise, endothelial blood cells damaged by high glucose levels, oxidative stress from excessive reactive oxygen species (ROS) formation, and chronic inflammation are more susceptible to atherosclerotic plaque formation and formation of arterial thrombus. Therefore, T2DM is often accompanied by cardiovascular disorders and a higher probability of stroke. It has been shown that T2DM increases risk of coronary heart disease (hazard ratio [HR] 2.00; 95% CI 1.83–2.19), ischaemic stroke (HR 2.27; 1.95–2.65), and other vascular disease-related deaths (HR 1.73; 1.51–1.98).
Recently, it was revealed that people with T2DM could develop impaired metabolic memory, which means there is a persistence of diabetic complications even after maintaining glycemic control. This suggestion appeared from the results of multiple large-scale clinical trials wherein complications could develop even with strict glycemic control is rigorous and special drugs after diabetes onset. These permanent metabolic abnormalities include aberrant gene expression in target organs/cells, an excess of ROS generation by mitochondria, chronic inflammation, endothelial cells dysfunction, vasoconstriction, and vasodilation disorders causing hypertension.
To date, the available medical therapies for the management of T2DM are directed on enhancing insulin secretion from the residual β-cells or target insulin resistance. Unfortunately, sulfonylureas and metformin have not been shown to have a durable effect on glycemic control with the advancing duration of disease and metabolic memory impairment. Therefore, there is a high need to search and develop new approaches for T2DM management, preventing disease complications and restoration of beta cell function.
In this context, mesenchymal stem cells are naturally produced by the body and possess many features useful for regenerative therapy of several disorders. These features include immunomodulatory activity, angiogenesis, promotion of endogenous stem cells proliferation and differentiation, anti-fibrosis and anti-apoptosis action. All these activities have been revealed and studied by many scientific groups. It was shown that MSCs provided all these features by secretion of various growth factors, cytokines, miRNAs and its paracrine action, or cell-to-cell communication, but not by the direct differentiation and engraftment into patients tissues.
The possible proposed mechanisms of MSCs therapy for improvement of β-cells functions and T2DM symptoms alleviation are similar. These include reducing systemic low grade inflammation characteristic for all T2DM patients, promoting angiogenesis and endothelial cells function restoration, and stimulating resident stem cells in the pancreatic duct to differentiate into islets. MSCs infusion has been shown to reduce insulin resistance in rat T2DM models by several ways: activating the insulin receptor substrate 1/protein kinase B signaling pathways; enhancing the glucose transporter type 4 (GLUT-4) expression in insulin target tissues; suppressing NLRP3 inflammasome-mediated inflammation; directing adipose tissue macrophages into an anti-inflammatory phenotype, and; regulating hepatic glucose metabolism. It could be suggested that MSCs may help to reverse abnormal metabolic memory to normal mode of functioning.
Thus, MSCs therapy could be effective for patients with T2DM. There are about 30 registered clinical trials for therapy of T2DM with MSCs and the results of some of them are already published where positive action of MSCs infusions on patients' state have been demonstrated.
Below some published clinical trial results are provided.
1. The prospective, randomized, single-blinded placebo-controlled study evaluates the efficacy and safety of autologous bone marrow-derived stem cell transplantation in 21 patients with T2DM. Patients in the intervention group received MSC infusion through a targeted approach, and after 12 weeks, a second dose of stem cells was administered through the antecubital vein. Nine out of the 11 (82%) patients in the intervention group achieved a reduction in insulin requirement by ≥50% from baseline while maintaining HbA1c U+304F7%, whereas the patients in the control group (p = 0.002) had none. The insulin requirement decreased by 66.7% in the intervention group from 42.0 IU per day to 14.0 IU per day, while the control group decreased by 32.1% from 40.5 IU per day to 27.5 IU per day at 12 months. The glucagon-stimulated C-peptide significantly increased in the treated group of patients compared to the control group.
2. The phase I/II, randomized placebo controlled study with a follow-up period of 36-months was conducted in 61 patients (42-63 years) diagnosed with T2DM according to the criteria outlined by the American Diabetes Association. Patients in intervention group I were administered two WJ-MSCs intravenous infusions within a four week interval. Patients in group II were treated with normal saline administered in the same volume of parenteral solution as WJ-MSC. All patients remained on the same drug therapy, diabetic diet, and exercise regimen as before. Following WJ-MSCs infusion therapy, a gradual decrease in HbA1c was detected in the patients in group I and the lowest level was registered at the sixth month of follow-up (baseline was 7.67±1.23%; at the 6 months 5.69±0.79%), after which HbA1c remained stable for 18 months. The results of other clinical trials demonstrated that WJ-MSCs was able to: improve the function of islet β-cells, as indicated by the increase in fasting C-peptide and HOMA-β; ameliorate hyperglycemia, as indicated by the decrease of FPG, PPG, HbA1c and the dosage of oral hypoglycemic agents and insulin therapy, and; reduce the incidence of diabetic complications.
3. A non-placebo controlled prospective phase I/II study was performed to determine efficacy and safety of WJ-MSC transplantation in 22 patients with T2DM (Chinese Clinical Trial Register ChiCTR-ONC-10000985). All patients received WJ-MSC transplantation through one intravenous injection and one intrapancreatic endovascular injection (catheterization). Follow up period was for 12 months after transplantation. MSCs transplantation was shown to decrease the levels of fasting glucose (baseline, 7.53 ± 2.67 mmol/L; 6 months after treatment, 7.12 ± 1.81 mmol/L; P U+304F0.05) and glycated hemoglobin (baseline, 8.20 ± 1.69%; at 3 months after treatment, 6.89 ± 0.90%; P U+3040.01); improved C-peptide levels (baseline, 1.29 ± 0.83 ng/mL; 6 months after treatment, 1.95 ± 1.3 ng/mL; P U+3040.05) and beta cell function, reduced markers of systemic inflammation and T lymphocyte counts. Insulin suspension occurred for 7 of 17 (41%) patients who received insulin after stem cell transplantation. 5 of 17 (29%) patients had a reduction in insulin requirement by ≥50%. In the remaining five patients, the dosage of insulin decreased to different degrees, except for one patient who was a non-responder. In total, one of five patients who received oral anti-diabetic drugs became completely drug-free three months after treatment and had good control of blood glucose with only diet and exercise intervention. No major WJ-MSC transplantation-related adverse events were reported.
3. A single-center, double-blinded, randomized, placebo-controlled phase II trial (NCT02302599) was performed to determine the efficacy and safety of umbilical cord-derived mesenchymal stem cells (UC-MSCs) in Chinese adults with type 2 diabetes mellitus (T2DM). 91 patients received intravenous infusion of UC-MSCs (n = 45) or placebo (n = 46) three times with 4-week intervals and followed up for 48 weeks. At 48 weeks following treatment, 20% of the patients in the UC-MSCs group achieved the level of HbA1c < 7.0% and daily insulin reduction ≥ 50%, compared with 4.55% in the placebo group (p < 0.05). Additionally, it was shown that UC-MSCs treatment reduced daily insulin requirement, decreased HbA1c levels, and ameliorated insulin resistance in a time-dependent manner. No major UC-MSCs transplantation-related adverse events were revealed. Thus, the potential efficacy and safety of UC-MSCs transplantation for the treatment of T2DM was concluded.
Overall, obesity, sedentary lifestyle, and excessive food intake, containing large amounts of fats and carbohydrates, often lead to T2DM development accompanied by hypertension and atherosclerosis. These metabolic disorders dramatically reduce the quality and duration of lifetime. Moreover, the number of adults aged between 20 and 79 years old living with diabetes will likely rise up to 700 million by 2045 according to the International Diabetes Federation (IDF). And the true disease burden of T2DM is possibly underestimated as 1 in 3 diabetic people who are underdiagnosed. Therefore, we highly recommend every three-six months monitoring of HbA1c and glucose levels in your blood, along with lipid profile.
Kintaro cells are bone marrow derived mesenchymal stem cells that possess perfect regenerative abilities for patients with T2DM, or could be used as prophylactic therapy to delay T2DM onset for genetically predisposed people. Together with moderate physical activity (for example walking for at least 20-40 min a day) as well as a healthy but calorie restricted diet, Kintaro cell therapy could help to prevent T2DM associated complications development and support active mode of lifestyle. The earlier hyperglycemia is revealed and treated, the more effective stem cell therapy will be.
Sources:
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7503727/
- https://journals.physiology.org/doi/full/10.1152/physrev.00045.2011
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5433043/
Links for clinical trials:
- https://journals.sagepub.com/doi/10.3727/096368913X665576?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4997981/#!po=41.6667
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4055092/
- https://stemcellres.biomedcentral.com/articles/10.1186/s13287-022-02848-6
