MESENCHYMAL STEM CELLS: New therapy for Autism Spectrum Disorder
Autism spectrum disorder (ASD) is a complex and heterogeneous neurodevelopmental disorder that often manifests in early childhood. According to the Centre of Disease Control and Prevention (CDC), the estimation of the overall prevalence of ASD in the United States was 16.8 per 1000 (or 1 in 59) for 8-year-old children in 2014. This disorder is four times more likely to occur in boys compared with girls, and the median age of children with ASD diagnosis was 52 months.
Generally, the main symptoms of ASD include repetitive behaviors, restricted range of activities, and impairments in social communication. These features may all be associated with a lack of integrity between the different brain regions that need for normal executive, social/emotional, and communicative behaviors. Given the complexity of this disorder, ASD could be divided into three conditions: (1) Asperger’s disorder, (2) autistic disorder, and (3) pervasive developmental disorder not otherwise specified (PDD-NOS).
In some cases, ASD could be present with other comorbidities like anxiety, depressive disorders, epilepsy, and hearing impairment. Due to ASD being a heterogeneous disorder, the causes of its development involve a complex interaction of multiple genetic and environmental risk factors, including some prenatal and postnatal drug exposures, immune dysregulation, and chronic neuroinflammation.
For genetic factors, several chromosomal abnormalities and candidate genes could be associated with ASD (for example, UBE3A, GABRB3, ENGRAILED 2 (EN2), SEROTONIN TRANSPORTER (5HTT), REELIN (RELN) and many others). Some of these candidate genes, including the major histocompatibility complex class genes, however, are needed for the immune system to work properly. Recent studies have also revealed altered DNA methylation in ASD, as well as an autoimmune disease in the family history increases the chance of ASD developing.
On the other hand, there are also several environmental factors that increase ASD development. These include the following :
- Maternal immune activation (MIA) due to infections (including measles, mumps, herpes, pneumonia and chickenpox);
- Various metabolic disorders associated with diabetes mellitus, hypertension, zinc deficiency, abnormal melatonin synthesis;
- Maternal mental health problems such as schizophrenia, anxiety, and depression that led to taking certain medications such as sodium valproate (used in treatment of epilepsy and bipolar disorder), some antidepressants, thalidomide, misoprostol, terbutaline;
- Early life exposure to stress, and;
- Advanced parental age.
Therefore, epigenetic mechanisms and specific environmental factors may serve as triggers for genetically predisposed persons to the development of ASD.
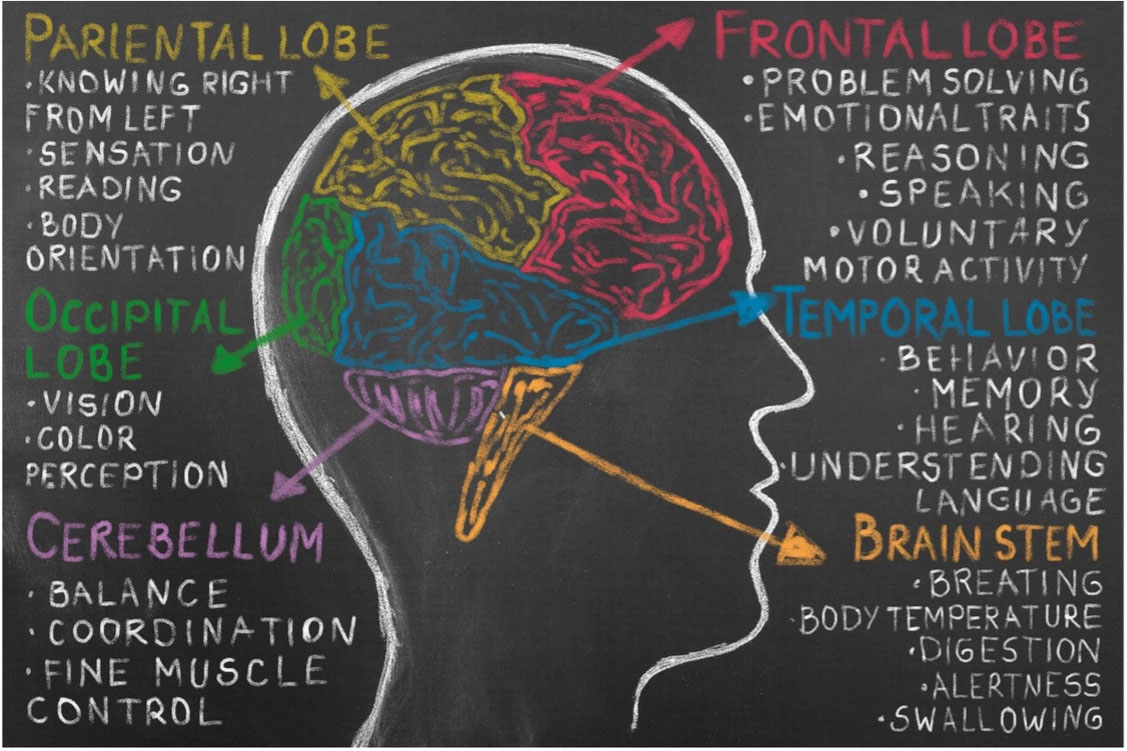
In that context, immune dysregulation with imbalance T- cell responses and elevated proinflammatory cytokine levels might be the starting point of ASD pathogenesis. Chronic up- images sources : https://www.psycom.net/autism-brain-differences regulation levels of proinflammatory cytokines in the cerebral spinal fluid and extreme microglial activation lead to abnormal axon connectivity, disruption of synapse function and plasticity, blood endothelial cells damaging and hypoperfusion. Brain hypoperfusion influences progenitor cell proliferation, neuronal differentiation and migration, axon and dendrite outgrowth, and synaptic function. All in all, these processes result in the lack of integrity between the different brain regions and visible symptoms of ASD.
That said, there is no one-fits-all treatment regimen for children with ASD. It is suggested that treatment should be started as early as possible, using a combination of speech, occupational, and behavioral therapies for training the skills needed to adapt to the conventional lifestyle. Clearly, there are no specific drugs approved for the treatment of ASD causes. Medications have only been used to relieve some of the associated symptoms. For example, fluoxetine and clomipramine were investigated in clinical trials for reducing repetitive behaviors. Methylphenidate, on the other hand, has been demonstrated to be effective in children with symptoms of hyperactivity and inattention.
Likewise, there is a close association between the nervous and immune system in early development. Neuroinflammation, microglial activation, and immune dysregulation have been suggested as the main features of many ASD cases. It has shown that there are correlations between proinflammatory cytokine levels and the severity of autistic symptoms. As a result, immunomodulatory therapies are of keen interest for ASD treatment.
One immunomodulatory therapy is the administration of mesenchymal stem cells (MSCs) to the patients' bodies. MSCs appeared effective for the treatment of several disorders associated with inflammation, immune system overactivation after organ or bone marrow transplantation, as well as autoimmune reactions.
In particular, MSCs act on both the adaptive and innate immune system by suppressing dendritic cell maturation, polarizing macrophages towards anti-inflammatory M2-like state, promoting the generation of regulatory T cells via IL-10, suppressing proliferation and cytotoxicity of NK cells, and reducing B cell activation and proliferation. MSCs have also been demonstrated to modulate neuroinflammation, provide neuroprotection, and enhance synaptic function by secretion of various growth factors. All these features make MSCs attractive candidates for therapy of certain neurologic conditions including ASD.
Cases Reports
- The patient was a 5-year-old boy that weighed 15 kg and was 98 cm tall. He was diagnosed with ASD at 18 months old. The stem cell therapy includes six intravenous injections of 1x106/kg umbilical cord MSCs, one dose every week. After the stem cell therapy, an improvement in verbal and nonverbal communication were demonstrated, and the patient was able to behave actively similar to the children of his age.
- The patient was a 6-year-old boy that weighed 17 kg and was 120 cm tall. Four intravenous injections of 1x106/kg umbilical cord MSCs were performed. After treatment, his relationship with people, as well as his emotional and physical response improved significantly. The patient was able to imitate other people, such as singing-along, and had progress in eye contact and in verbal and nonverbal communication. Currently, the patient is attending a general elementary school as his interpersonal relationships greatly improved.
- The patient was 4-years old with a confirmed diagnosis of autism. Magnetic resonance imaging of the brain showed no significant intracranial abnormality and electroencephalogram was normal in the sleep state. The child was non- verbal prior to stem cell therapy. A score of 142, representing an autism index >99%, was estimated using Gilliam Autism Rating Scale 2 (GARS-2) assessment instrument for the stereotypical behavior and the social interaction subscale. The therapy included 3 intrathecal administrations of autologous bone marrow aspirate concentrate (BMAC). Additionally, the patient has occupational and psychological therapy, sensory integration, and speech therapy. Major improvements were revealed in body use, intellectual response, visual response score, taste, smell, touch score and fear or nervous score. The child started to speak and a score of 91 has been estimated according to GARS-2, representing an autism index of 27%. Thus, significant improvements were observed compared with the previous score ( GARD-2 score 142, autism index > 99%).
Both of the patients were able to build a sentence with 4-5 words in comparison to hardly using 1-2 words prior to the umbilical cord MSCs treatment.
Clinical trials
Single-arm phase I/II clinical trial with repeated doses of intravenously administered umbilical cord mesenchymal stem for 20 children with ASD was performed by Riordan et al. (NCT02192749). MSCs were infused in a dose of 36 million every 12 weeks (four times over a 9-month period) and were followed up at 3 and 12 months after treatment completion. These infusions appeared safe and generally well tolerated. Forty percent of children (eight persons) showed notable improvements of symptoms as measured by the Autism Treatment Evaluation Checklist (ATEC) and the Childhood Autism Rating Scale (CARS), placing them in a lower ASD symptom category when compared with baseline prior to the therapy started. Levels of macrophage-derived chemokine (MDC) and activation-regulated chemokine (TARC) levels also decreased for five of these eight children. The mean MDC, TARC, ATEC, and CARS values achieved their lowest levels, 3 months after the last infusion.
Similarly, the open-label, phase I study of MSCs therapy for 12 children with ASD between 4 and 9 years of age was performed by Sun et al. (NCT03099239). Therapy included intravenous (IV) infusions of unrelated donor umbilical cord tissue mesenchymal stromal cells (hCT-MSCs). Children received one, two, or three doses of 2 × 106 cells per kilogram at 2-month intervals. The therapy was well tolerated, and the side effects were minimal. Clinical and laboratory effectiveness evaluations were performed at baseline, 6 months and 12 months after the final infusion. Improvement in core symptoms of ASD were described in 50% of participants.
On the other hand, Sharma et al. investigated the effects of autologous bone marrow mononuclear cells (BMMNCs) intrathecal transplantation in 32 patients with ASD. The study included children and adults with ASD (age 3-33 years). Most of the patients showed improved scores in various behavioral scales after a 26 month of follow up. It has been reported that insufficient blood flow in the brain occurs in many brain regions in ASD and the cell transplantation was suggested to have a balancing effect on the brain metabolism. Tomography- Computed Tomography (PET-CT) scans before and 6 months after cell transplantation showed increased 18F-fluorodeoxyglucose uptake in the areas of frontal lobe, cerebellum, amygdala, hippocampus, parahippocampus, and mesial temporal lobe. Out of 32 patients, a total of 29 (91%) patients improved in total ISAA scores and 20 patients (62%) showed decreased severity on CGI-I. On CGI- II, 96% of patients showed global improvement. Few adverse events in this study such as seizures in three patients were controlled with medications.
Another similar small open label study investigated the clinical benefits of bone marrow aspirate concentrate stem cell with intrathecal transplantation in 10 ASD children (4-12 years of age). The maximal effect of cell therapy was observed within the first 12 months following the treatment. An interesting and very important observation of this study was the decreasing efficacy of stem cell therapy as the age of child with ASD increases. In other words, the functional changes of the brain with ASD accumulated with age appear less amenable to treatment and recovery with MSC therapy.
At present, there are small amounts of published data concerning ASD therapy with MSCs. All these studies showed that therapy with MSCs is safe, resulting in only mild or moderate adverse effects, which are short in duration. There is no current consensus regarding the optimal dosage or frequency of MSC administration, but studies are in concordance that repeated doses of MSCs are more beneficial than a single dose for the treatment of ASD. The earlier the therapy starts, the more positive effects from therapy with MSCs could be experienced.
Suggested MSCs mechanism of action:
- Modulation of the immune system, leading to reduction of neuroinflammation and proinflammatory cytokines production, and inhibition of microglial cells activation.
- Improvement of neural connectivity by new synapse formation.
- Reduction of brain hypoxia caused by hypoperfusion and blood vessels damages.
- Stimulation of endogenous neural progenitor cells by various growth factors.
- Balance of the brain metabolism.
Overall, MSCs therapy can improve the social relationships, emotional responsiveness, speech, language, communication, behavior patterns, sensory aspects, and cognition in some ASD patients. Kintaro cells suggest multiple doses of bone marrow-derived MSCs obtained from young healthy donors for the therapy of ASD. Our MSCs therapy appeared safe and effective for children with ASD, the same to the reported clinical trials and case reports above.
Sources:
- https://www.europeanreview.org/wp/wp-content/uploads/8075-8080.pdf
- https://stemcellsjournals.onlinelibrary.wiley.com/doi/full/10.1002/sctm.19-0434
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6397804/
- https://link.springer.com/article/10.1007/s12015-021-10257-0
- https://www.hindawi.com/journals/sci/2013/623875/
- https://academic.oup.com/stcltm/article/8/10/1008/6403779?login=false
- https://ejnpn.springeropen.com/articles/10.1186/s41983-022-00525-2
- https://academic.oup.com/stcltm/article/9/10/1137/6406967
- http://jscholaronline.org/full-text/JCROA/5_106/A-Case-Study-on-Autism-Spectrum.php
